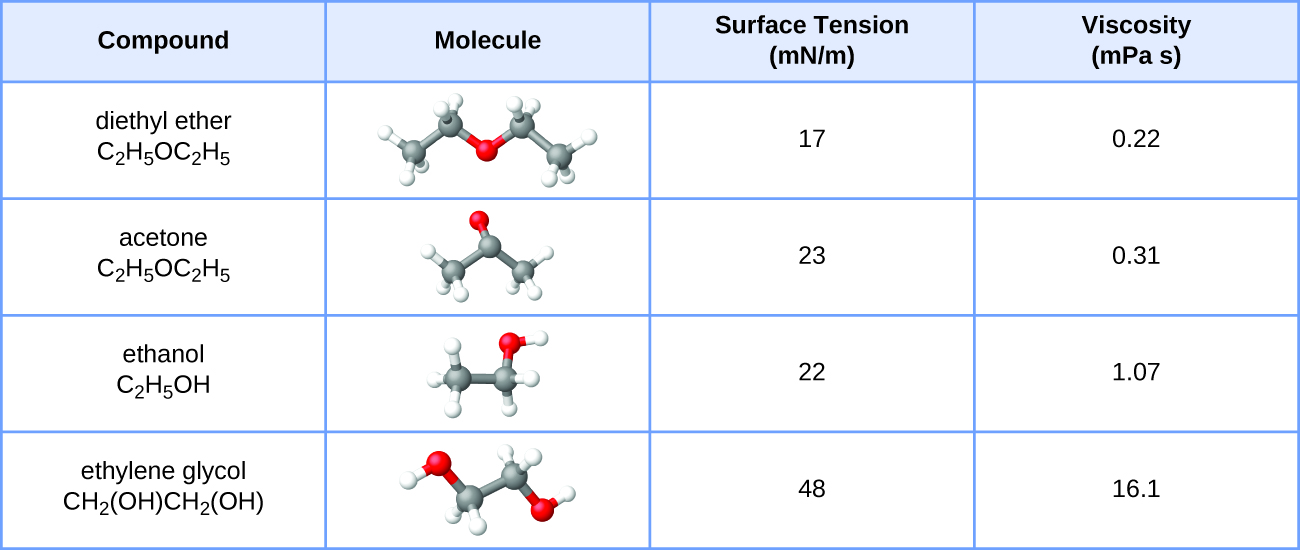
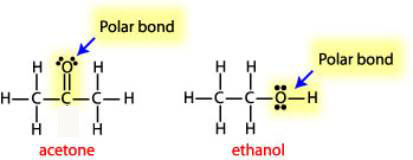
Draw the structures of ethanol, acetone, toluene, hexane, and water. Classify each solvent as polar, nonpolar, or moderately polar. | Homework.Study.com

Key Roles of Lewis Acid–Base Pairs on ZnxZryOz in Direct Ethanol/Acetone to Isobutene Conversion | Journal of the American Chemical Society

Combining acetone-butanol-ethanol production and methyl orange decolorization in wastewater by fermentation with solid food waste as substrate - ScienceDirect
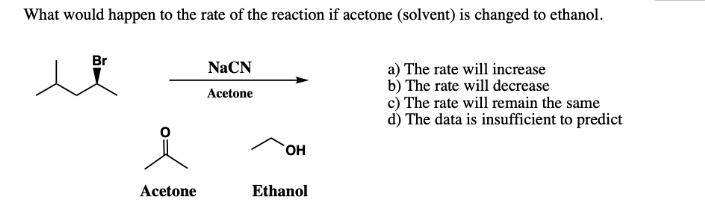
SOLVED: What would happen to the rate of the reaction if acetone (solvent) is changed to ethanol. NaCN Acctone a) The rate will increase b) The rate will decrease The rate will

thermodynamics - What mixing ratio of ethanol and acetone has the lowest freezing point? - Chemistry Stack Exchange
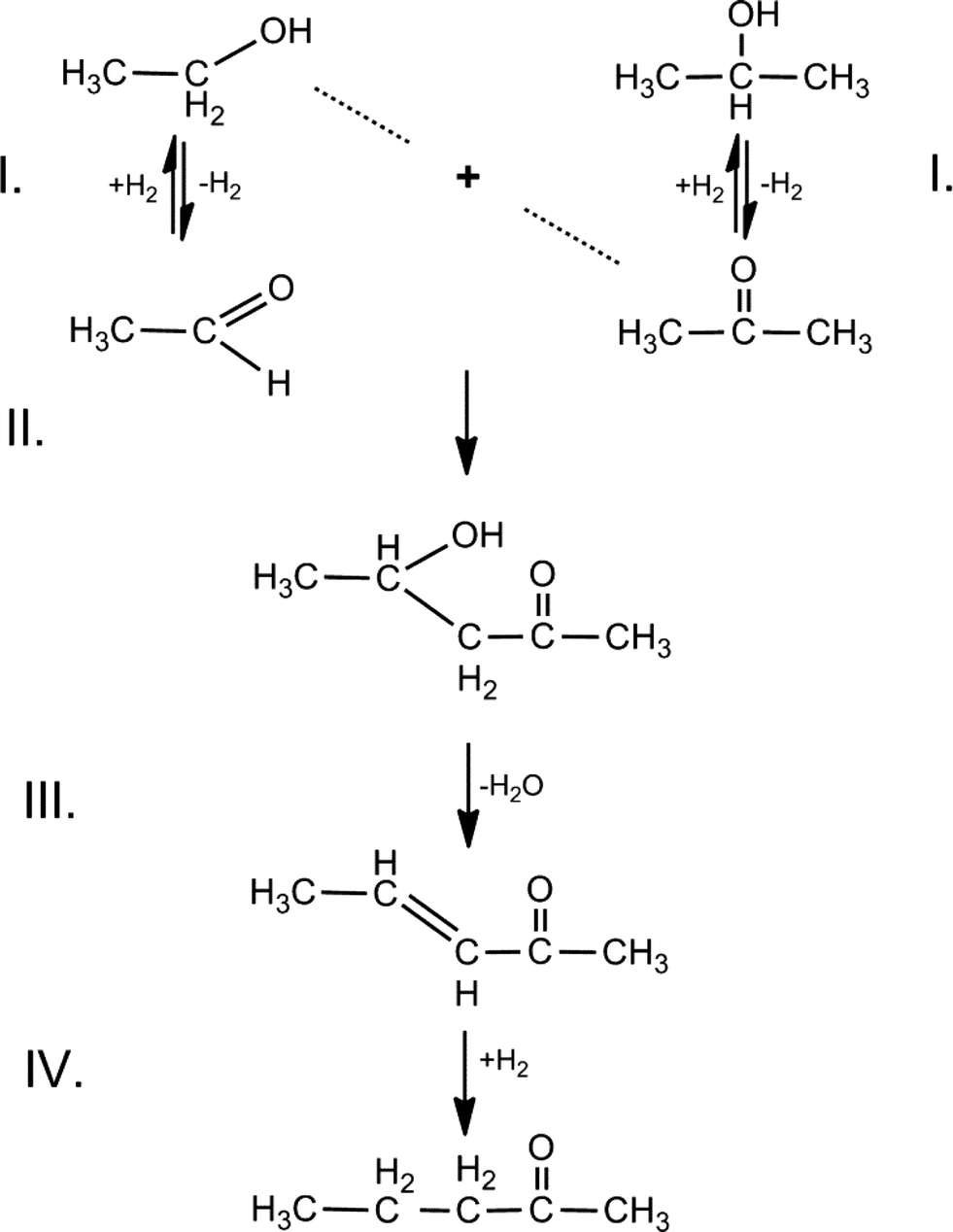
Upgraded biofuel from alcohol–acetone feedstocks over a two-stage flow-through catalytic system - Catalysis Science & Technology (RSC Publishing) DOI:10.1039/C6CY00025H

Mechanism and Kinetics of Isobutene Formation from Ethanol and Acetone over ZnxZryOz | ACS Catalysis

Structure formula and size of acetone, ethanol, and water, considering... | Download Scientific Diagram

Acetone is treated with excess of ethanol in the presence of hydrochloric acid. The product obtained is: