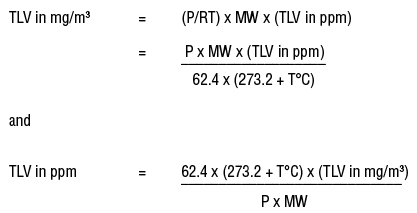
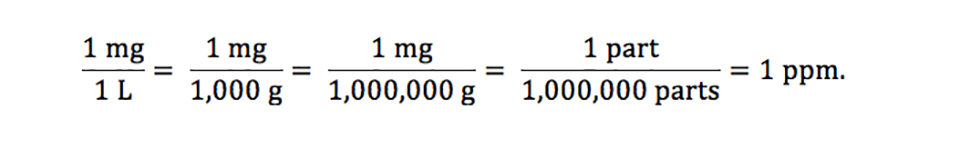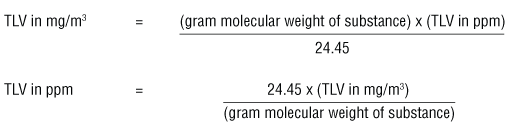SOLVED: Calculating the Water Hardness For convenience assume that all hardness is due to the presence of only Caz+ions in the form of calcium carbonate, CaCO3 The unit of hardness may be

Concentration of solutions Parts per million (ppm) This is a way of expressing very dilute concentrations of substances. Just as per cent means out of. - ppt download