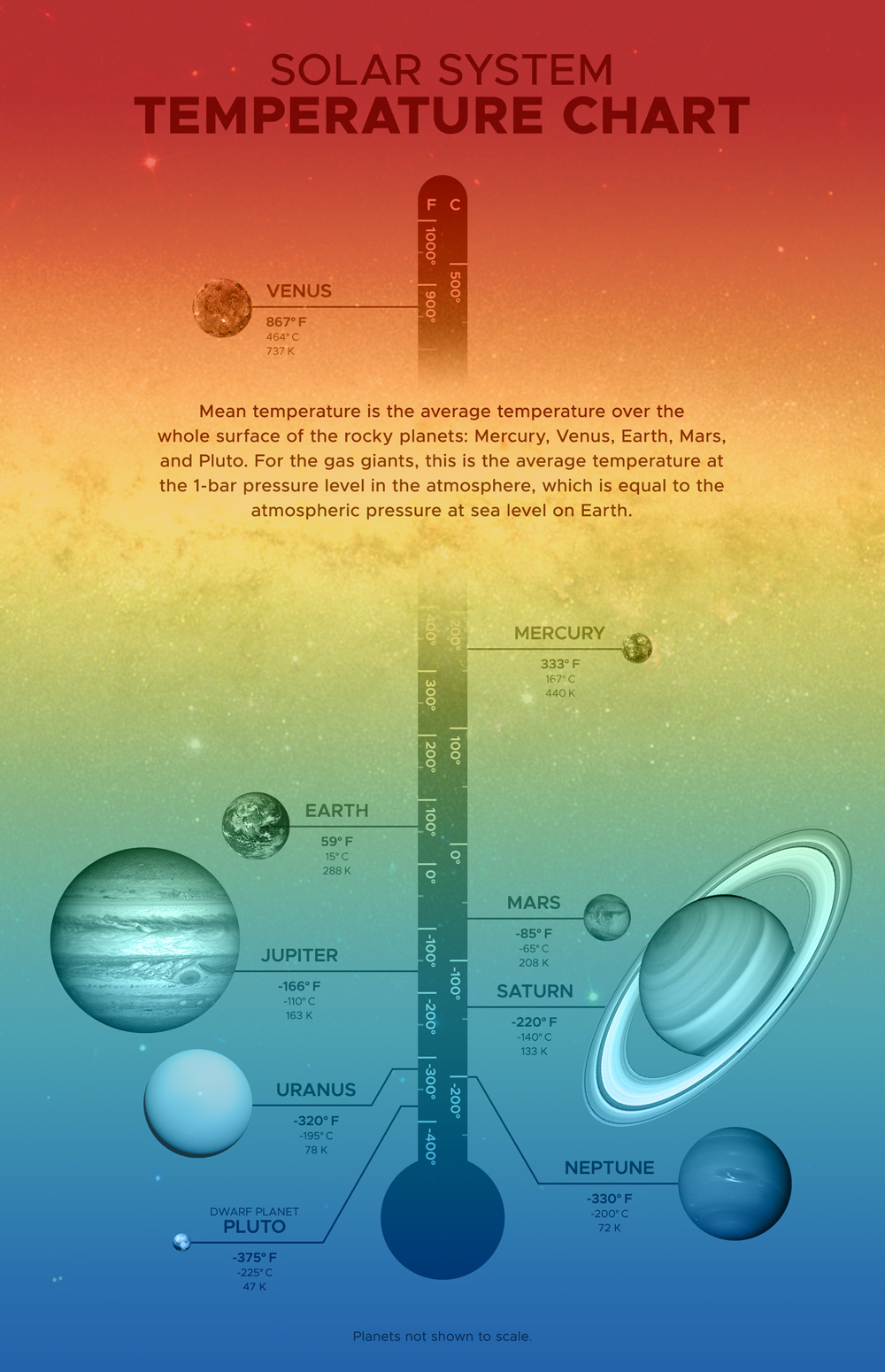
The solubility of iodine in water is 1.1 xx 10^(-3) "mol L"^(-1) at 288 K. When 0.200 g of iodine is stirred in 100 of water till equilibrium is reached, what will

Langmuir isotherms plots for the adsorption of 5BIS on carbon steel (a)... | Download Scientific Diagram

SOLVED:The atmosphere becomes colder at higher elevation. As an average the standard atmospheric absolute temperature can be expressed as Tatm= 288-6.5 ×10^-3 z, where z is the elevation in meters. How cold

In a calorimeter of water equivalent 20g ,water of mass 1.1 kg is taken at 288K temperature.If steam at temperature 373K is passed through it and temperature of water increases by 6.5^oC

Fitting results of q 2 (a) under 288 K, (b) under 298 K, and (c) under... | Download Scientific Diagram

c p E Correlation of the system acetone (1)+dodecane (2) at 288 K, with... | Download Scientific Diagram

In a calorimeter of water equivalent 20g ,water of mass 1.1 kg is taken at 288K temperature.If steam at temperature 373K is passed through it and temperature of water increases by 6.5^oC
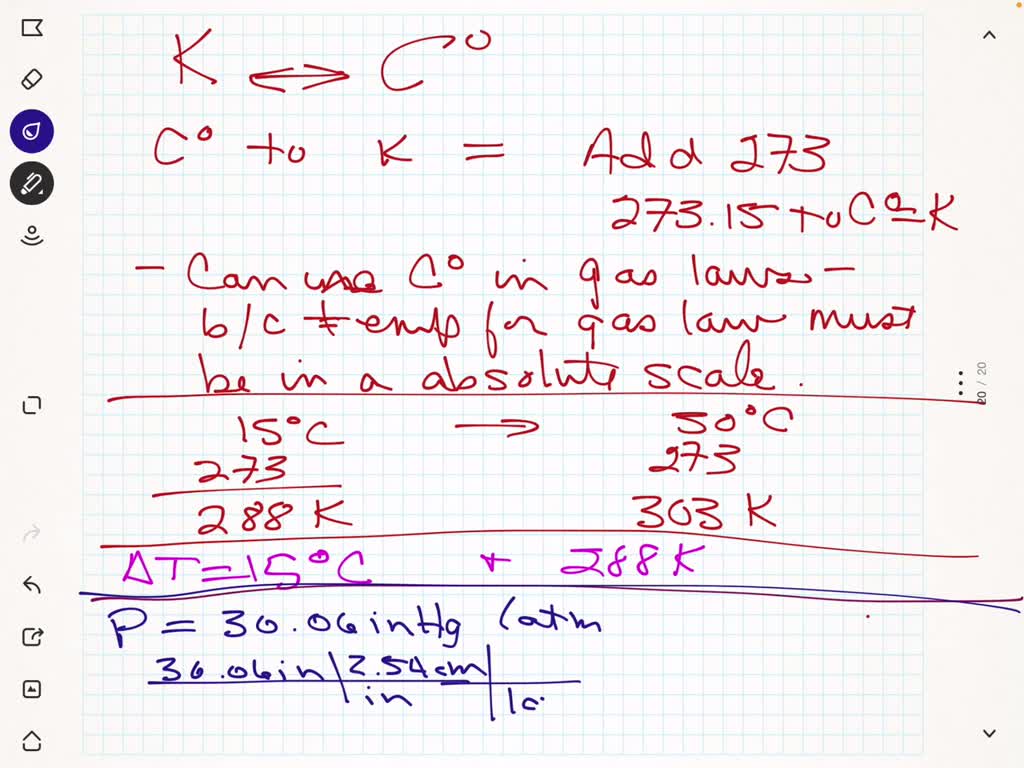
SOLVED: What is the conversion from Celsius to Kelvin and why is this conversion useful when applying gas law calculations? A room is heated from 15 °C to 30 °C. What are

Figure 11 | CuO Nanoparticle-Protein Bioconjugate: Characterization of CuO Nanoparticles for the Study of the Interaction and Dynamic of Energy Transfer with Bovine Serum Albumin | SpringerLink