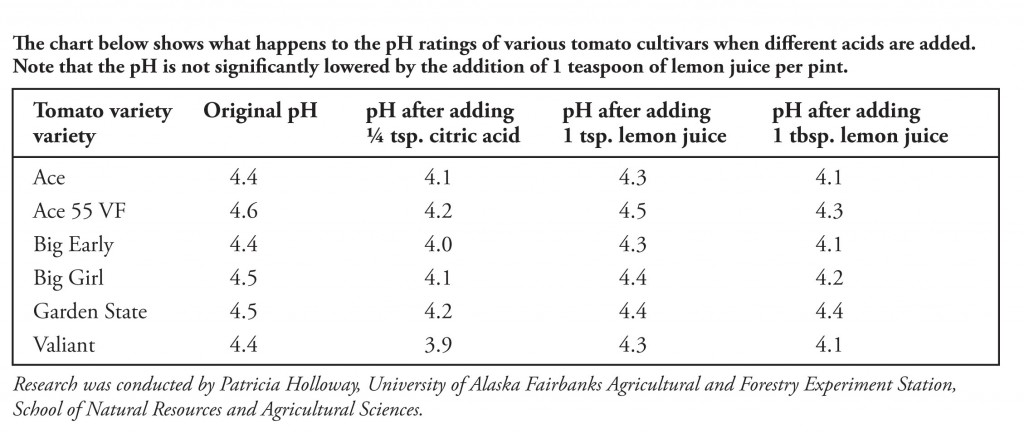
Lemon juice normally has a `pH` of `2`. If all the acid the lemon juice is citric acid and there are - YouTube

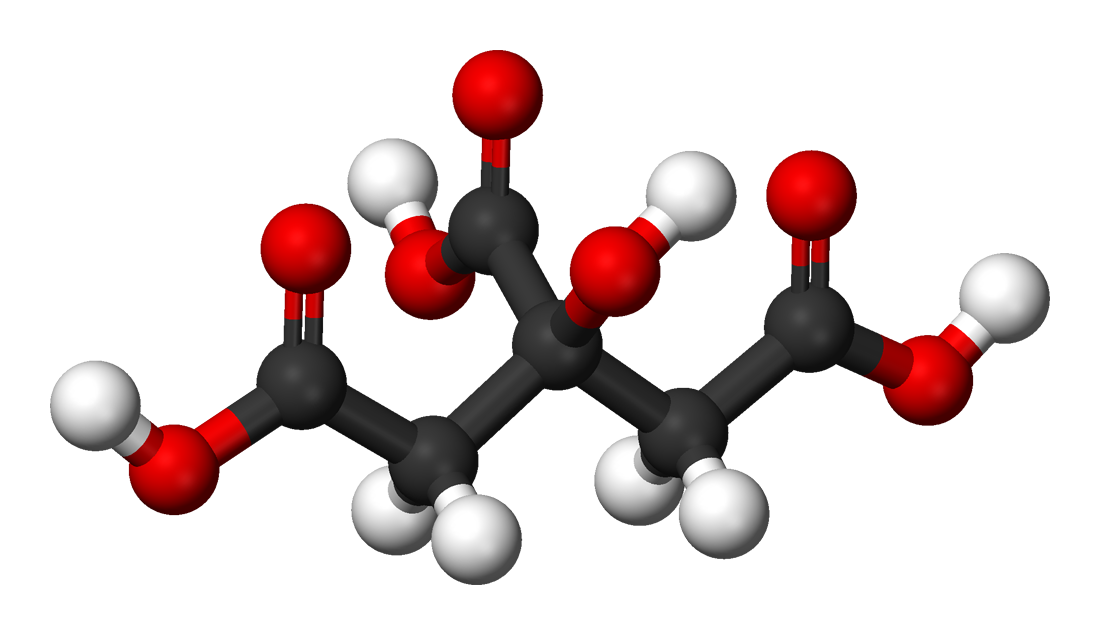
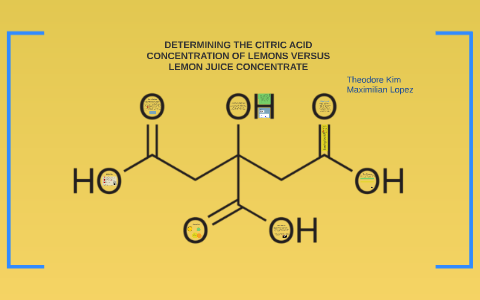
Lemon juice contains citric acid with a concentration of approximately 0.30 M (around 6% by mass), giving rise to its acid properties. Citric acid is generally considered a triprotic acid (capable of

Lemon juice has a pH = 2.1 . If all the acid in lemon is citric acid (H "Cit." hArr H^(+)+"Cit"^(-1)) and K(a) for citric acid is 8.4xx10^(-4) mole/litre, what is the
Quantitative assessment of citric acid in lemon juice, lime juice, and commercially-available fruit juice products - Document - Gale OneFile: Health and Medicine

Determination of Citric acid in Soft drinks, Juice drinks and Energy drinks using Titration | Semantic Scholar

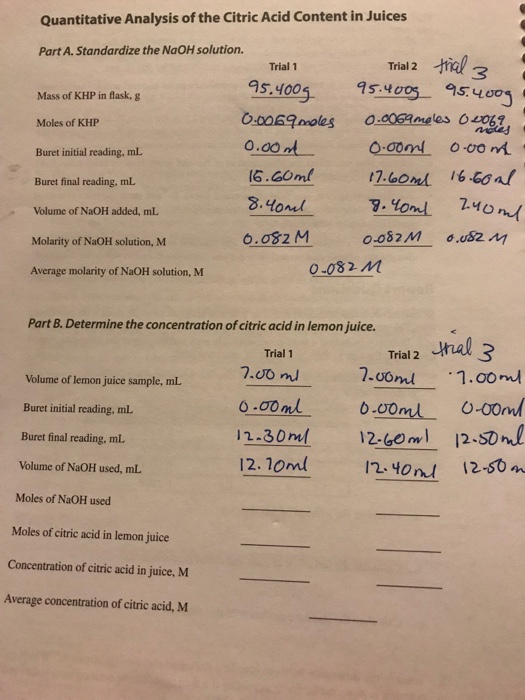
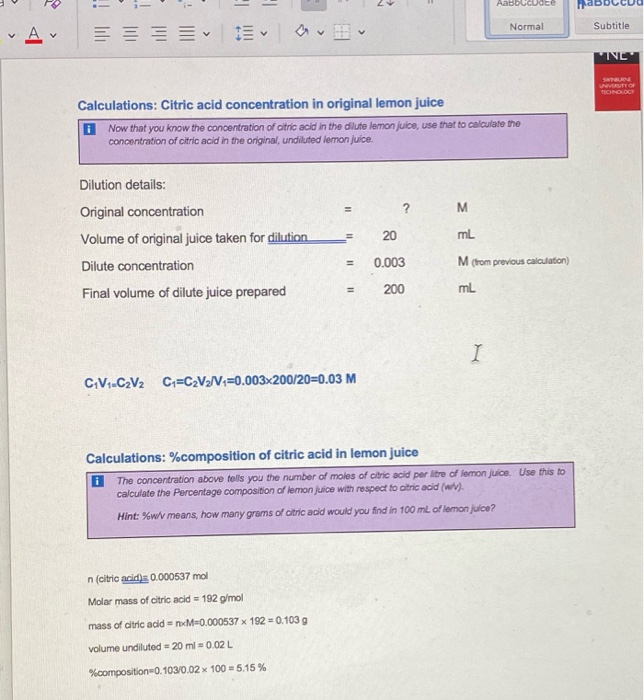




![PDF] Determination of Citric acid in Soft drinks, Juice drinks and Energy drinks using Titration PDF] Determination of Citric acid in Soft drinks, Juice drinks and Energy drinks using Titration](https://www.researchgate.net/profile/Eid_Brima/publication/264159629/figure/tbl1/AS:669163056013329@1536552313454/Citric-acid-concentrations-in-soft-drinks-Juice-drinks-and-energy-drinks_Q320.jpg)