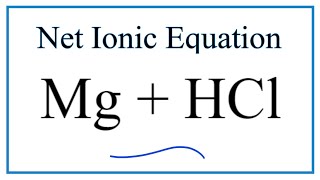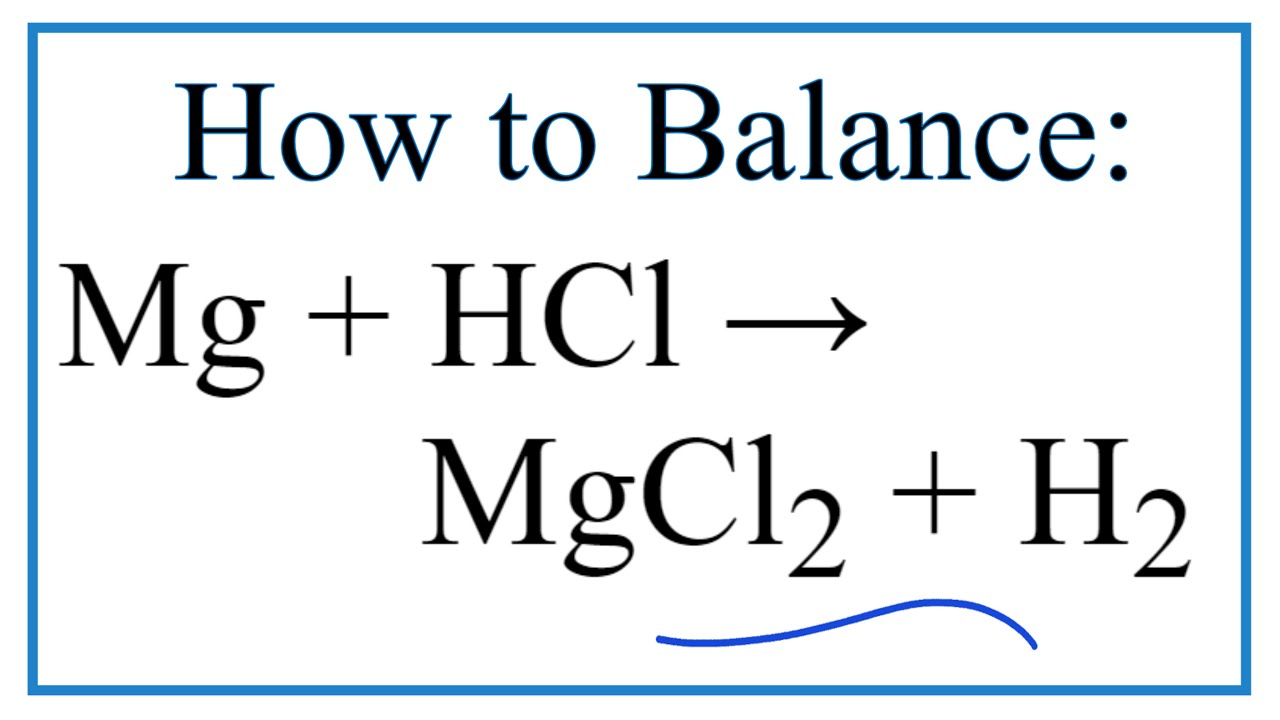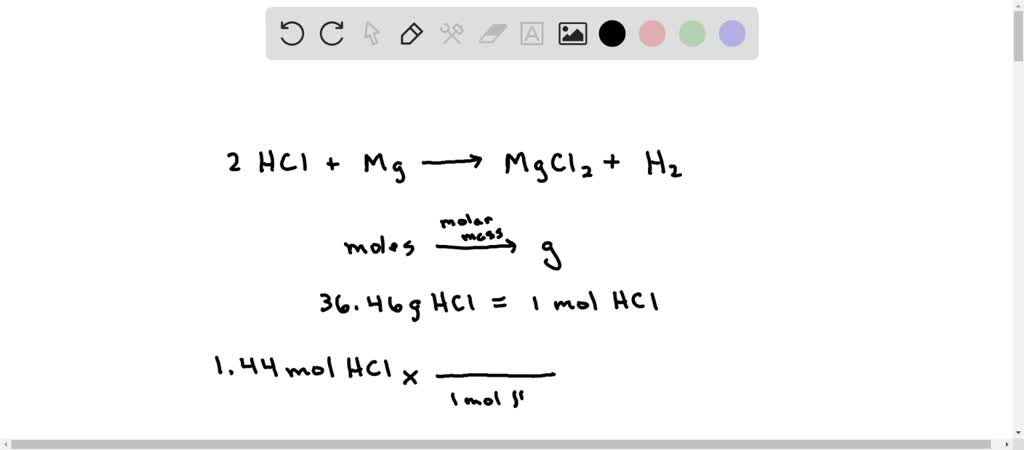
SOLVED: When magnesium metal (Mg) reacts with hydrochloric acid (HCl), magnesium chloride (MgCl2) and hydrogen gas (H2) are produced according to the following chemical equation. 2 HCl (aq) + Mg (s) →

What happens magnesium react with hydrochloric acid? - Find 3 Answers & Solutions | LearnPick Resources
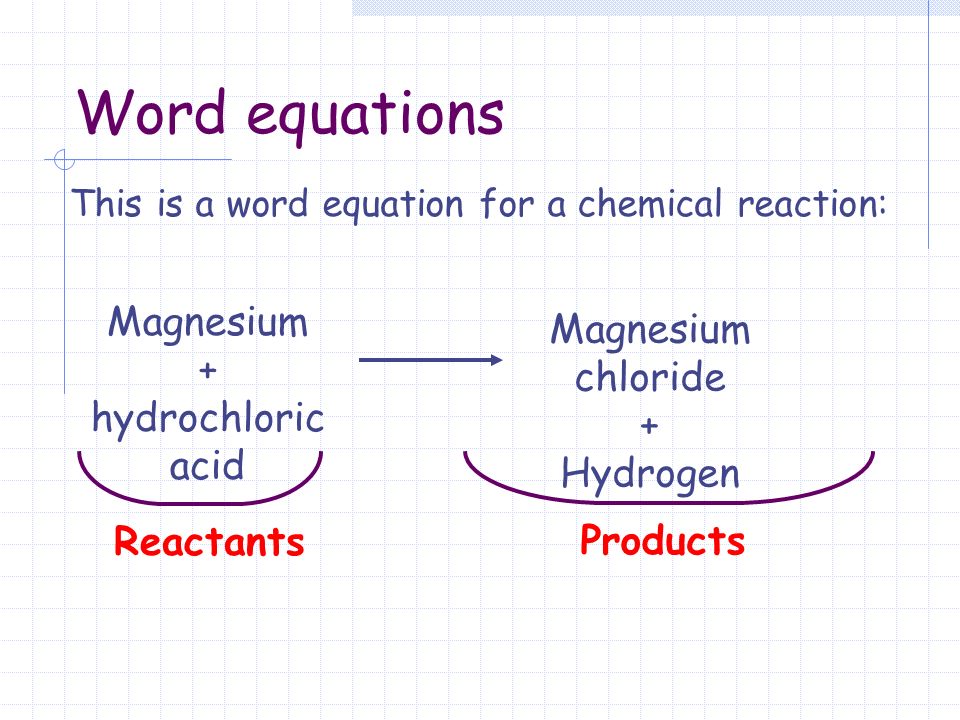
This is a word equation for a chemical reaction: Reactants Products Magnesium + hydrochloric acid Magnesium chloride + Hydrogen Word equations. - ppt download

investigate reactions between dilute hydrochloric and sulfuric acids and metals (e.g. magnesium, zinc and iron) Diagram | Quizlet
Exampro 2017 Page 1 NB This document may have been edited and can only be used in your school 4-5 Energy Changes – Chem

Describe an experiment to study the speed of reaction between dilute hydrochloric acid and magnesium, by measuring the volume of gas produced over time. - Study notes, tips, worksheets, exam papers

SOLVED: When hydrochloric acid reacts with magnesium hydroxide, the products that form are magnesium chloride and water. What is the coefficient for water when this equation is properly balanced?

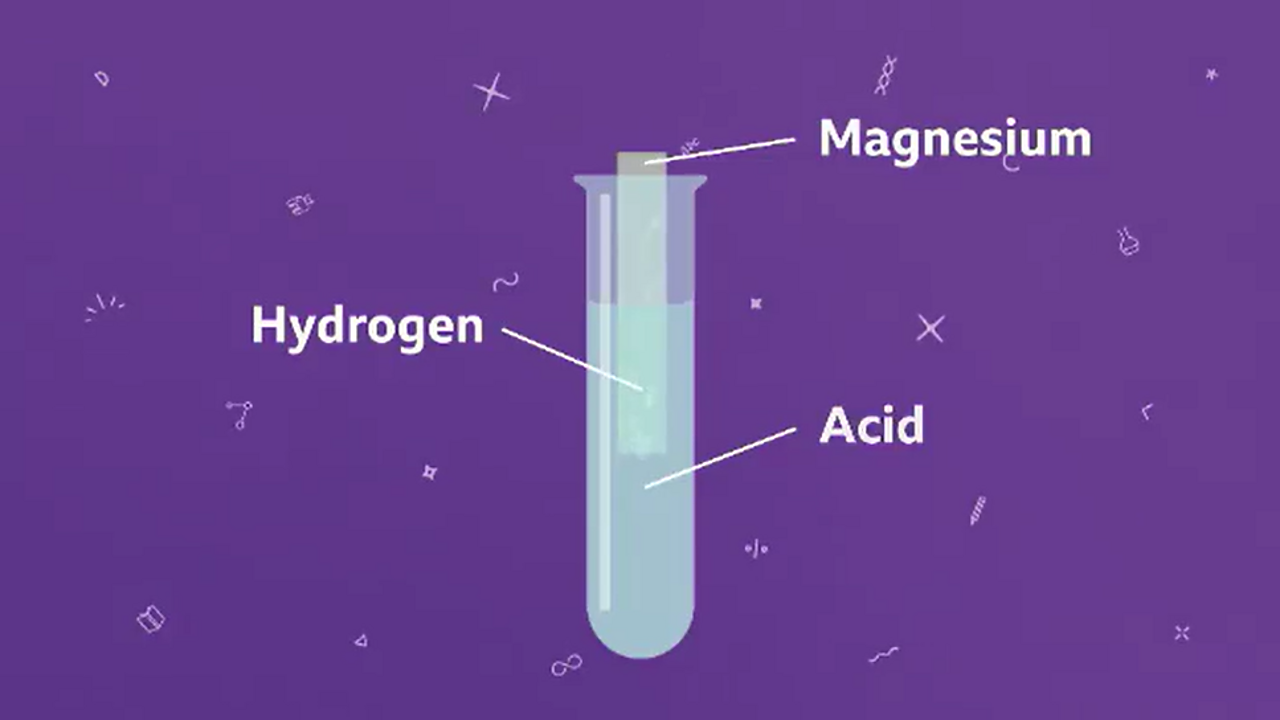
Question Video: Identifying the Name of the Gas Produced When Magnesium Metal Reacts with Hydrochloric Acid | Nagwa

Question Video: Recognizing Why Warm Hydrochloric Acid Has a Faster Rate of Reaction with Magnesium than Cold Hydrochloric Acid | Nagwa