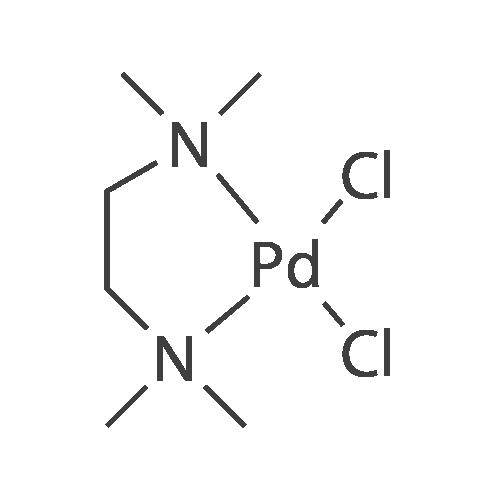
China Dichloro(N,N,N′,N′-tetramethylethylenediamine)palladium(II) CAS NO: 14267-08-4 Manufacturers - Free Sample - Alfa Chemical
![Palladium-mediated synthesis and biological evaluation of C-10b substituted Dihydropyrrolo[1,2-b]isoquinolines as antileishmanial agents - ScienceDirect Palladium-mediated synthesis and biological evaluation of C-10b substituted Dihydropyrrolo[1,2-b]isoquinolines as antileishmanial agents - ScienceDirect](https://ars.els-cdn.com/content/image/1-s2.0-S022352342100307X-ga1.jpg)
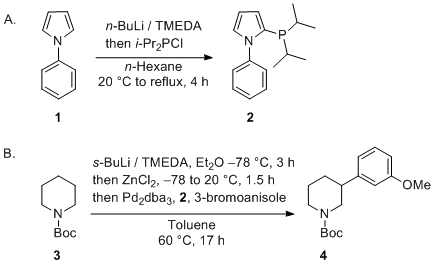
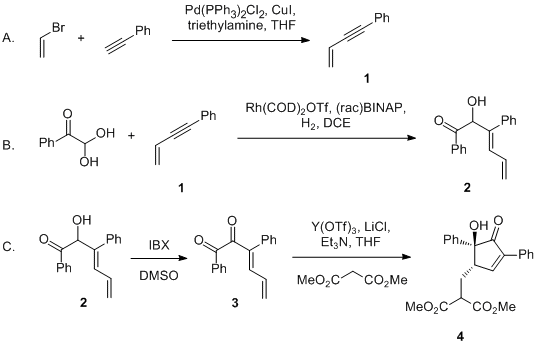
Palladium-mediated synthesis and biological evaluation of C-10b substituted Dihydropyrrolo[1,2-b]isoquinolines as antileishmanial agents - ScienceDirect

China Dichloro(N,N,N′,N′-tetramethylethylenediamine)palladium(II) CAS NO: 14267-08-4 Manufacturers - Free Sample - Alfa Chemical

A general synthesis of aromatic amides via palladium-catalyzed direct aminocarbonylation of aryl chlorides - Organic Chemistry Frontiers (RSC Publishing) DOI:10.1039/D2QO00251E

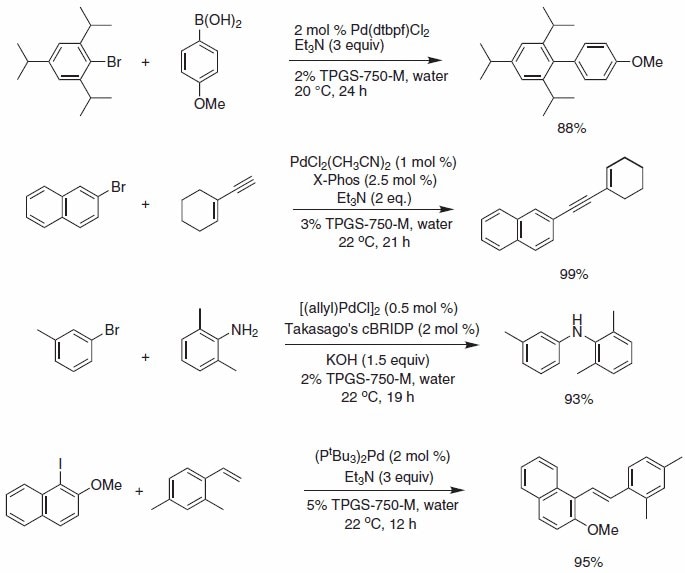
Sequential Unsymmetrical Aryl Coupling of o-Substituted Aryl Iodides with o-Bromophenols and Reaction with Olefins: Palladium-Catalyzed Synthesis of 6H-Dibenzopyran Derivatives | Organic Letters
![Synthesis of Mercury(II) and Palladium(II) Organometallic Derivatives of Terephthaldehyde by Metalation, Transmetalation, Oxidation, and Condensation Reactions. X-ray Crystal Structures of trans-[PdCl{C6H3(CO2H)2-2,5}(PPh3)2]·Me2CO·2MeOH, trans-[PdCl ... Synthesis of Mercury(II) and Palladium(II) Organometallic Derivatives of Terephthaldehyde by Metalation, Transmetalation, Oxidation, and Condensation Reactions. X-ray Crystal Structures of trans-[PdCl{C6H3(CO2H)2-2,5}(PPh3)2]·Me2CO·2MeOH, trans-[PdCl ...](https://pubs.acs.org/cms/10.1021/om970583u/asset/images/om970583u.social.jpeg_v03)
Synthesis of Mercury(II) and Palladium(II) Organometallic Derivatives of Terephthaldehyde by Metalation, Transmetalation, Oxidation, and Condensation Reactions. X-ray Crystal Structures of trans-[PdCl{C6H3(CO2H)2-2,5}(PPh3)2]·Me2CO·2MeOH, trans-[PdCl ...

NaBH4-TMEDA and a palladium catalyst as efficient regio- and chemoselective system for the hydrodehalogenation of halogenated heterocycles - ScienceDirect

Insertion of Molecular Oxygen into a Palladium(II) Methyl Bond: A Radical Chain Mechanism Involving Palladium(III) Intermediates | Journal of the American Chemical Society
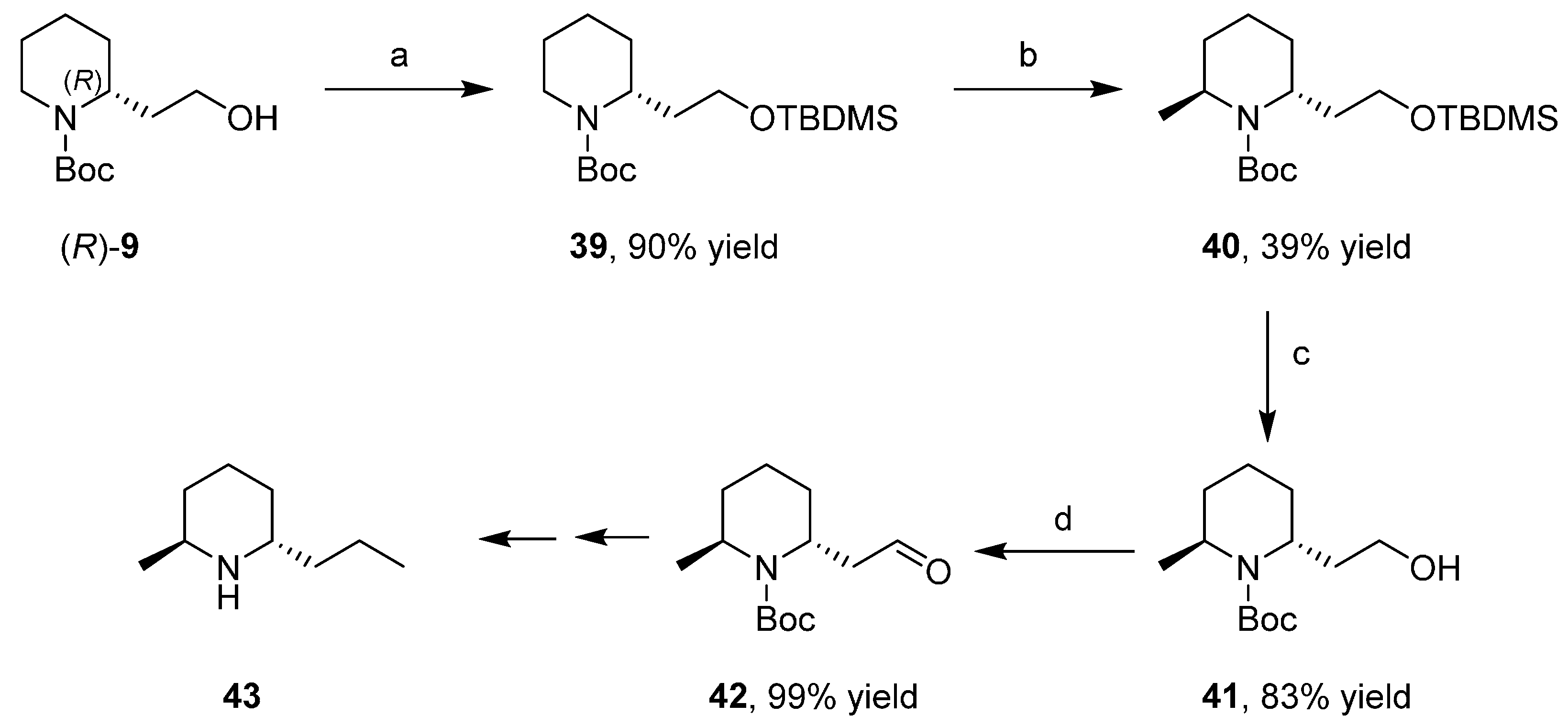
IJMS | Free Full-Text | Enzymatic Kinetic Resolution of 2-Piperidineethanol for the Enantioselective Targeted and Diversity Oriented Synthesis
![A Practical Synthesis of 6-[2-(2,5-Dimethoxyphenyl)ethyl]-4-ethylquinazoline and the Art of Removing Palladium from the Products of Pd-Catalyzed Reactions | Organic Process Research & Development A Practical Synthesis of 6-[2-(2,5-Dimethoxyphenyl)ethyl]-4-ethylquinazoline and the Art of Removing Palladium from the Products of Pd-Catalyzed Reactions | Organic Process Research & Development](https://pubs.acs.org/cms/10.1021/op034072x/asset/images/large/op034072xn00001.jpeg)
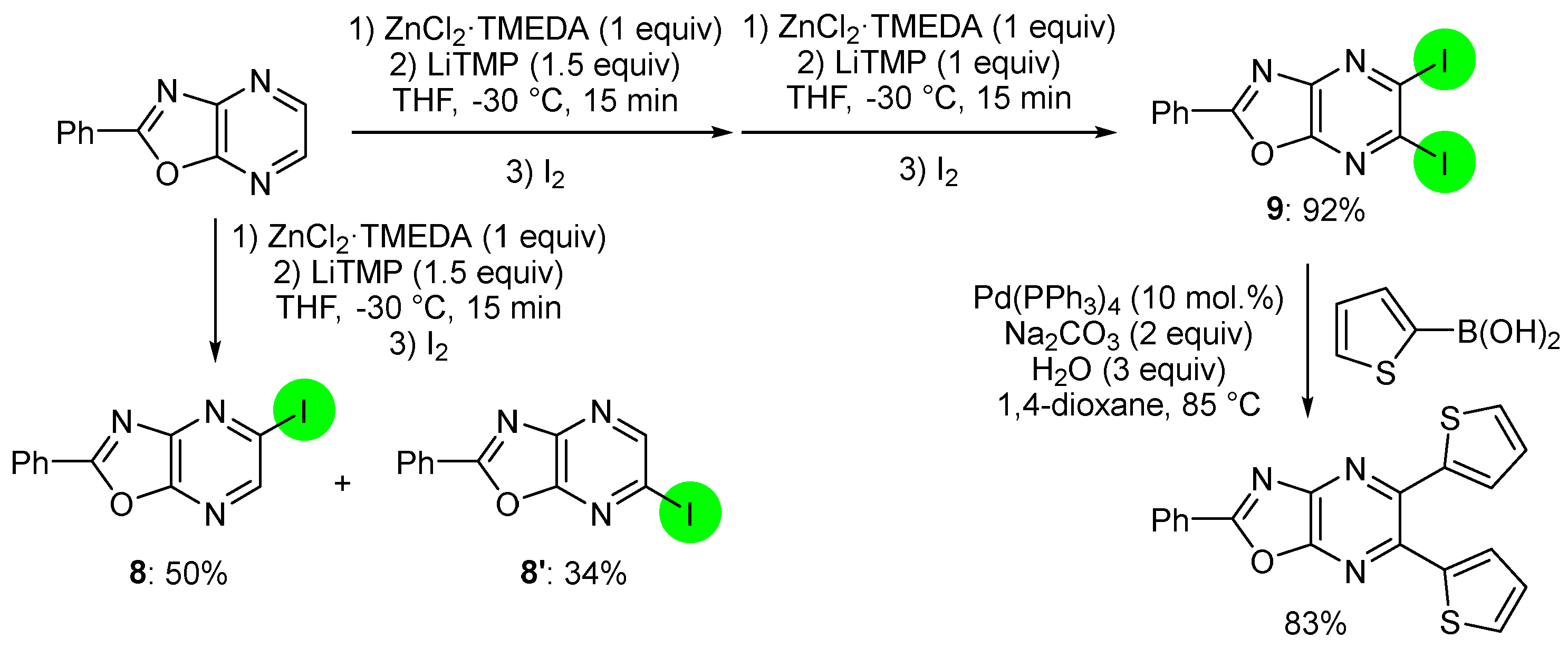
![Scheme 1. Synthesis of 2 from 1 and [Pd(dba)2]. | Download Scientific Diagram Scheme 1. Synthesis of 2 from 1 and [Pd(dba)2]. | Download Scientific Diagram](https://www.researchgate.net/publication/355315820/figure/fig1/AS:1080535237562368@1634631084024/Scheme-1-Synthesis-of-2-from-1-and-Pddba2.png)