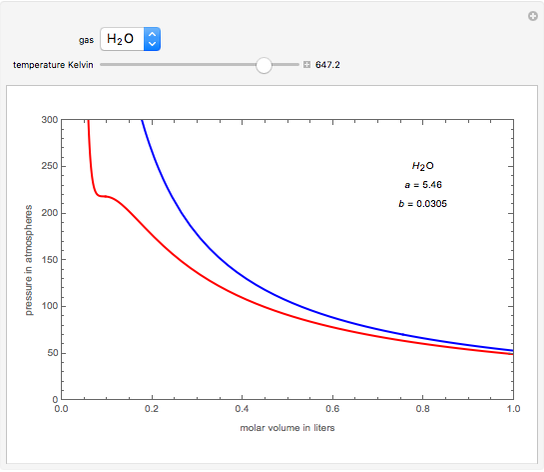
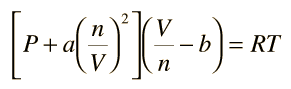The virial form of van der Waal's gas equation is PV = RT (1 + BV + CV^2 + .... ) = RT(1 + B'P + C'P^2 + ....) . The second

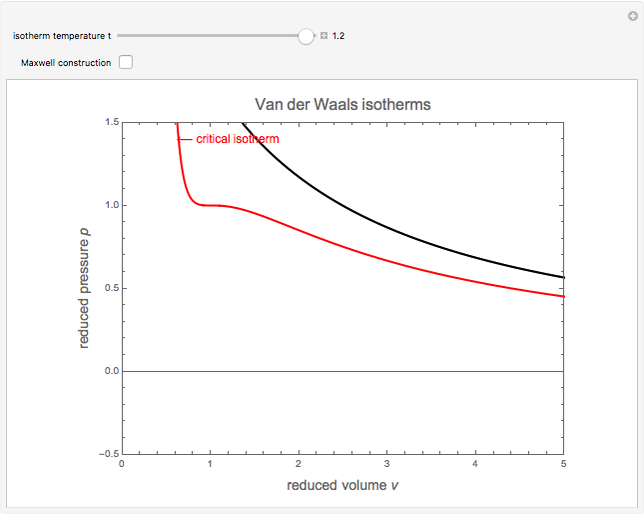
1. isothermal curves for the van der Waals EOS (it is extended by +∞... | Download Scientific Diagram

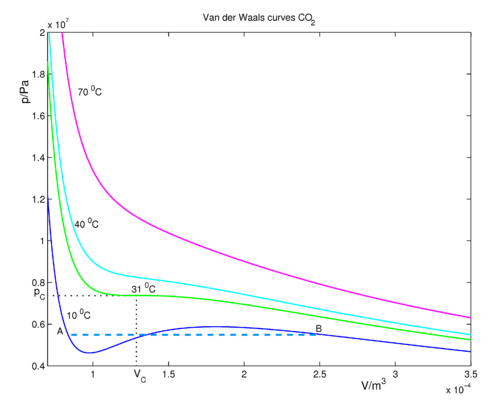
p-v diagram for a van der Waals gas with c = 1.0125. The variables are... | Download Scientific Diagram

Find the entropy increment of one mole of a Van der Waals gas due to the isothermal variation of volume from V1 to V2. The Van der Walls corrections are assumed to

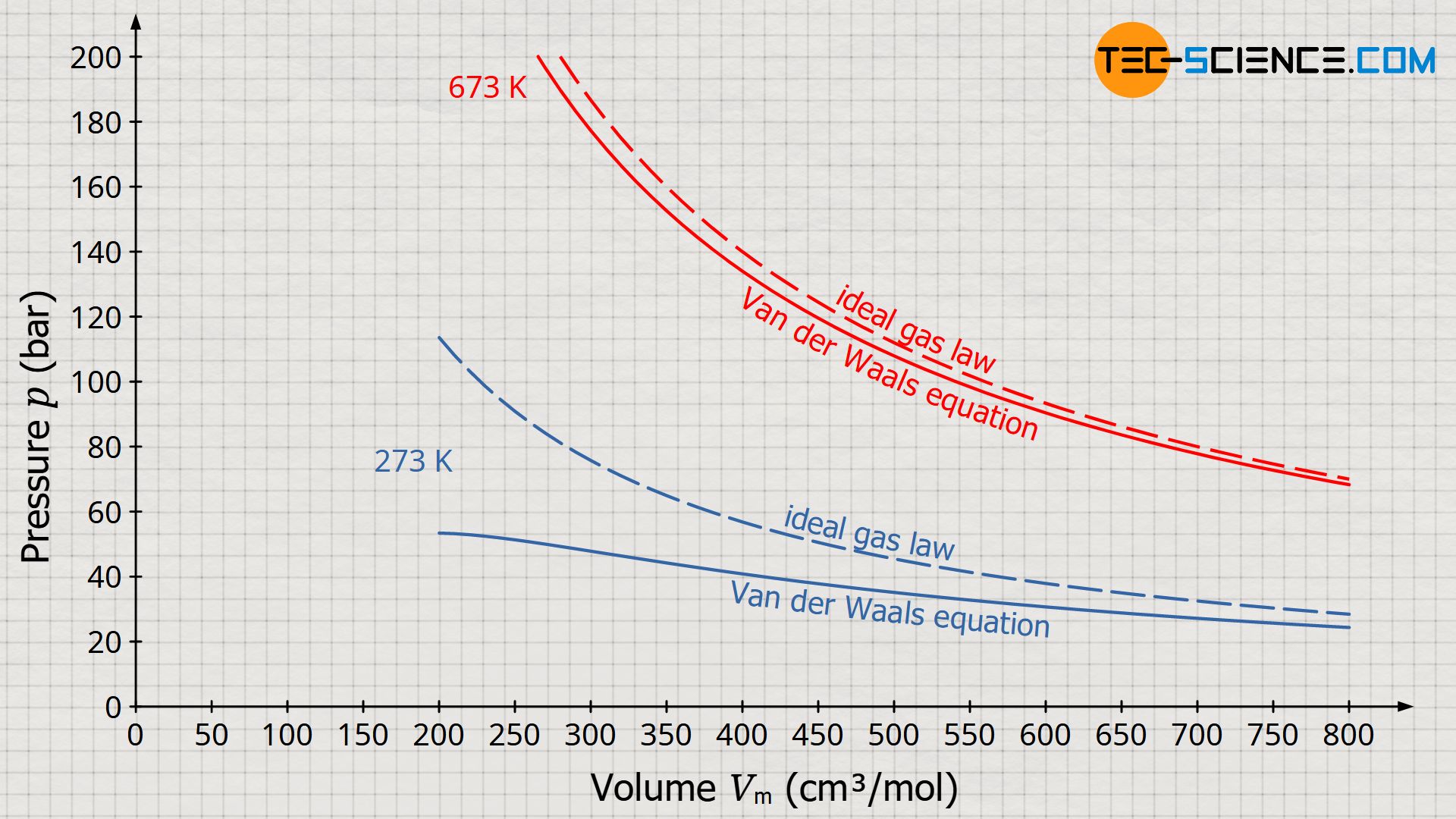
From van der Waals to VTPR: The systematic improvement of the van der Waals equation of state - ScienceDirect