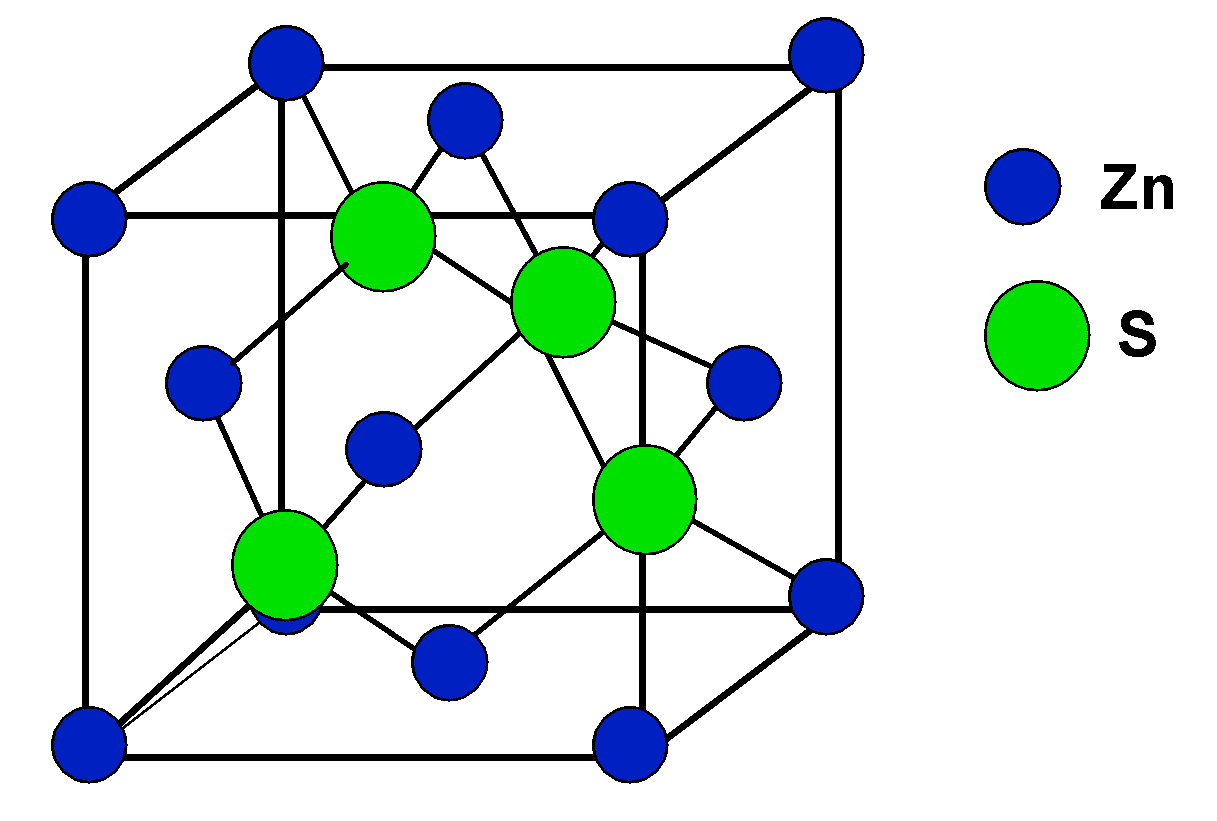
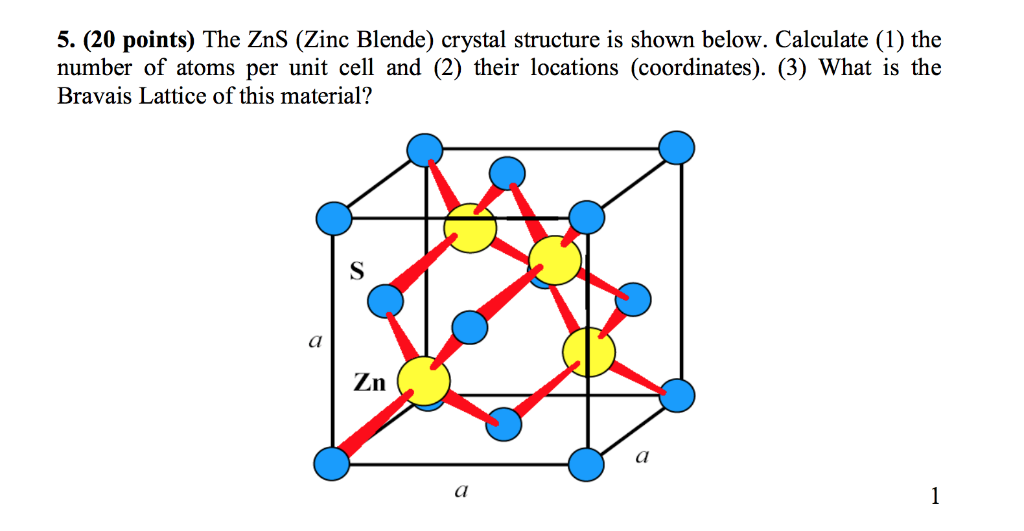
The compound $\\text{ CuCl }$ has a zinc blende structure and the edge length of its unit cell is 500 pm, its density (in $\\text{ g c}{{\\text{m}}^{\\text{3}-}}\\text{ }$ ) is :(Given that

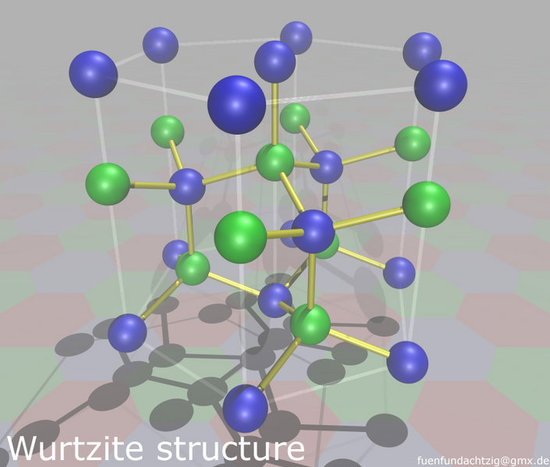
Zinc Blende structure - Wurtzite structure , arrangement of unit cell, packing fraction, of zinc blende structure with FAQs
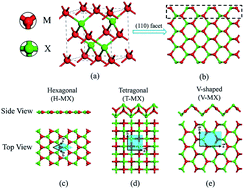
New manifold two-dimensional single-layer structures of zinc-blende compounds - Journal of Materials Chemistry A (RSC Publishing)
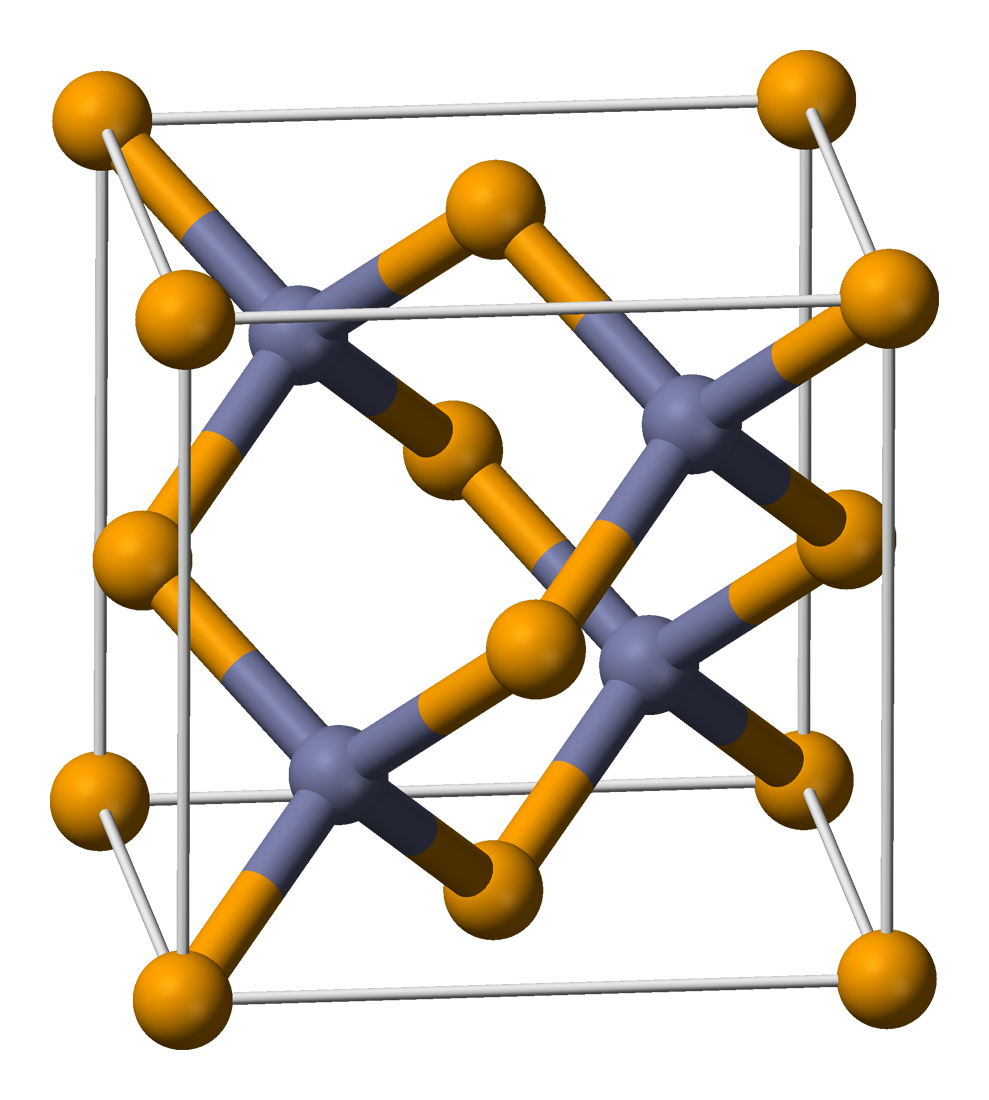
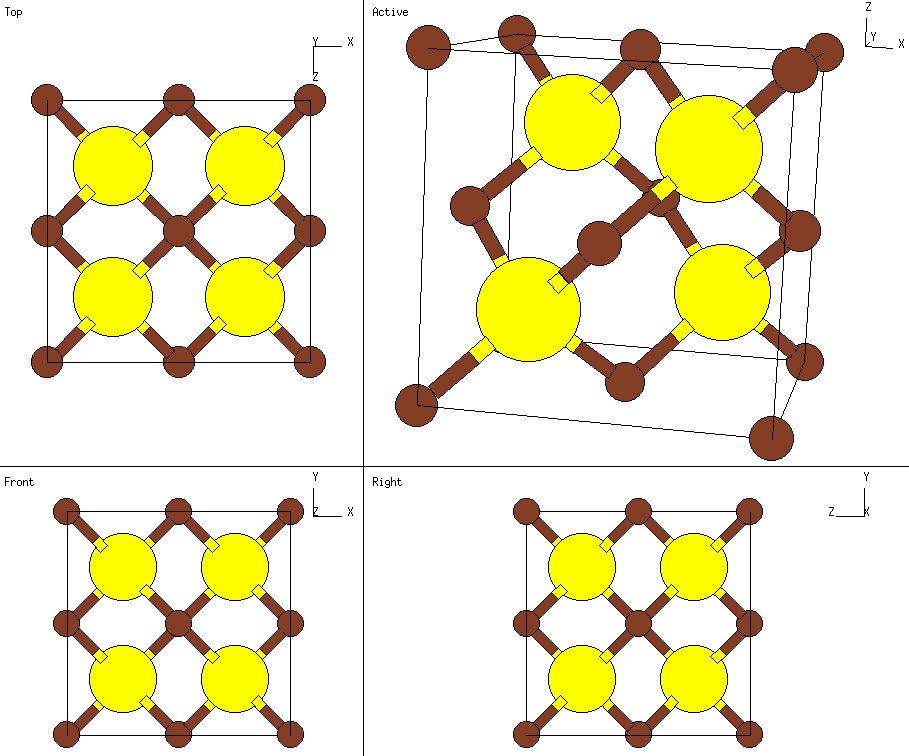
Polymorphism of bulk GaP and ZnS. (a) The zinc-blende structure with... | Download Scientific Diagram

Zinc Blende structure - Wurtzite structure , arrangement of unit cell, packing fraction, of zinc blende structure with FAQs

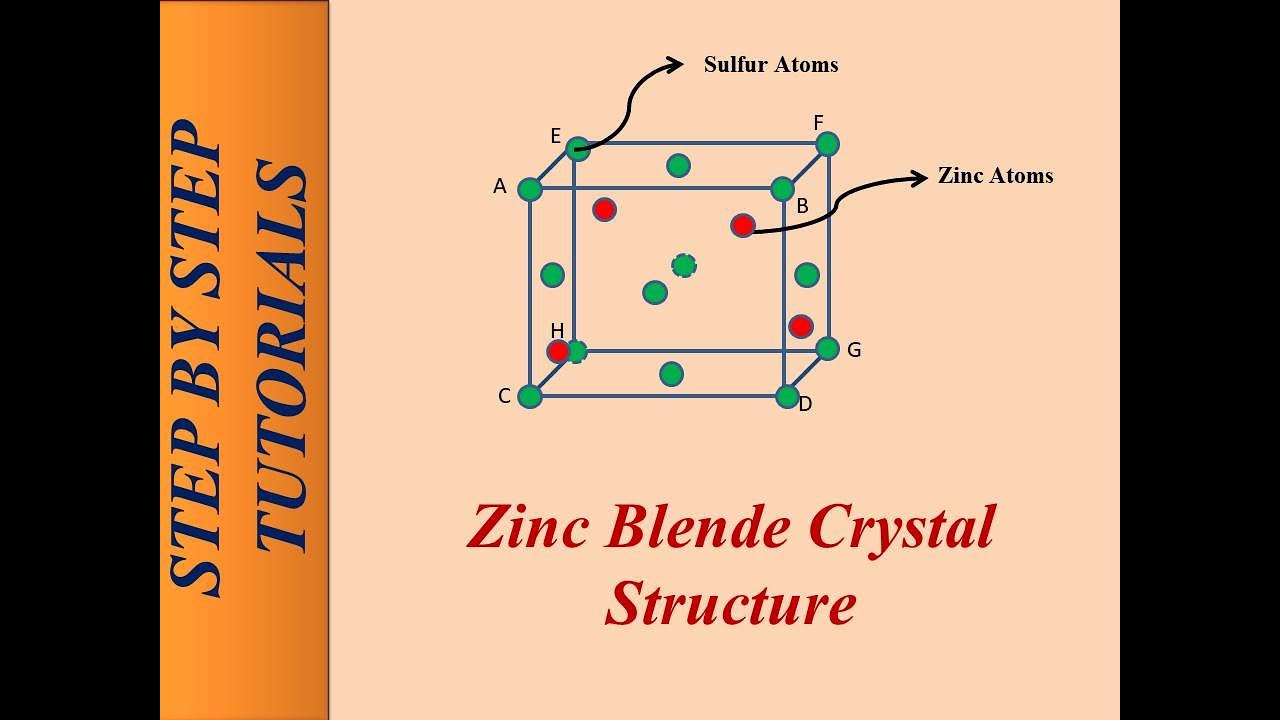






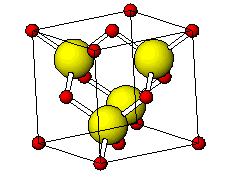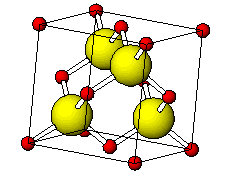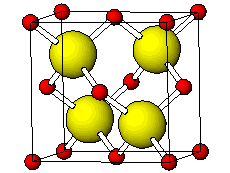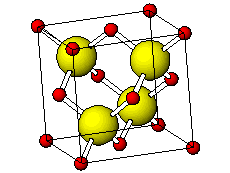
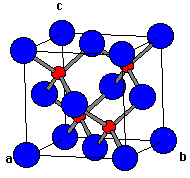

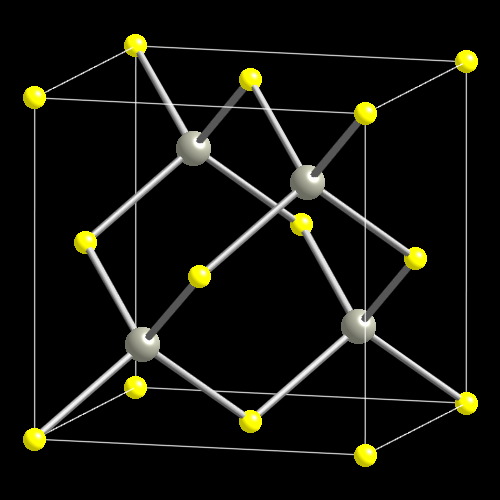
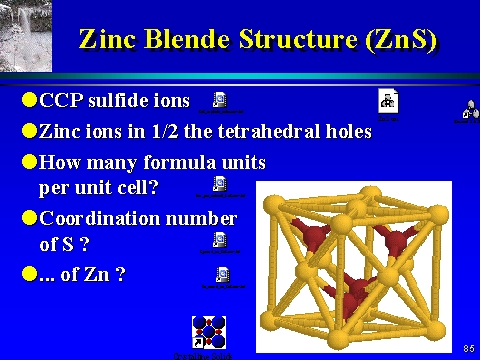
![PDF] Investigations of Phonons in Zinc Blende and Wurtzite by Raman Spectroscopy | Semantic Scholar PDF] Investigations of Phonons in Zinc Blende and Wurtzite by Raman Spectroscopy | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/ab5ac14082b211fcb78739d13a402101e865cd61/3-Figure1-1.png)